Your opinion
Other Languages
Español
Français
Interlingua
Português
|
|
ECOPOESIS.
Summary - The Ecopoesis
model is a hypothesis about the origin of life
on Earth that proposes that the geochemical cycles
of biogenic elements (or biogeochemical cycles)
preceded the appearance of organisms. The existence
of such cycles in a prebiological world implies
the presence of strong electron donors (reductants)
and acceptors (oxidants), replenished by abiotic
reactions in the earth's primitive aqueous environment.
Cycling of carbon, nitrogen and sulfur compounds
would take place in the oceans, an aqueous environment
in contact with a lithosphere rich in reducing
ions such as ferrous (divalent) iron and sulphide,
and an atmosphere containing substantial amounts
of molecular (free) oxygen, generated by the atmospheric
photolysis of water vapour.
The redox transitions pertaining to living processes
would thus be primordial attributes of the nascent
ecosphere, determining the nature of materials
and energy-yielding processes that would gradually
shape the evolution of organismality.
High atmospheric levels of carbon dioxide would
account for some essential reactive properties
of this protobiological world, such as the possibility
of carbon fixation, and chiral propagation.
The Ecopoesis model (Félix de Sousa, R.A.,
2000, 2006) for the origin of life is based on
the idea that an atmosphere containing a high
percentage of molecular oxygen, generated by the
atmospheric photolysis of water vapour, is an
essential feature of the Earth's earliest ecosphere.
Ecopoesis offers a different perspective as compared
with other models of biopoesis. Traditionally,
the origin of life is equalled to the fortunate
appearance of very simple cellular organisms,
whose metabolic evolution would thenceforth conduct
the general evolution of the environment (the
oxygen-rich atmosphere being a result of this
process). In ecopoesis, in contrast, it is the
physical environment that plays the leading role,
and not organisms.
Besides endowing the environment, from the very
beginning, with the continuous protection of an
ozone layer, the early presence of free oxygen
determines the onset of a flow of electrons, which
leaving the reducing components of the lithosphere,
cross the archean seas, pumped by the photolytic
production of oxygen in the Earth's atmosphere.
This flow is mediated by, and predominantly channelled
through the redox transitions of the redox-sensitive
elements in the hydrosphere.
These large-scale environmental interactions cause
the development of a geochemically based metabolism
in a planetary protoplasm (holoplasm) setting
the stage for the gradual evolution of organismal
life to take place.
|
The wide difference
of electrochemical potentials in the primordial
environment would cause the appearance of the geochemical
cycles of biogenic elements. The primitive metabolic
pathways are originated by the interaction of these
cycles and their products. The buildup of order
in the system arises from the energetically favourable
transitions, particularly in the oxidation of organic
matter, and from the physicochemical properties
of the compounds involved. This planetary protometabolism
is essentially congruent to today's biochemistry,
including carbon and nitrogen fixation, and aerobic
degradation of organic compounds (full oxidation
to CO2).
Biological evolution, as a rule, would proceed through
the increasing functional control of such reactions,
rather than through their creation. A naked geochemical
metabolism would thus evolve congruently towards
our modern enzymatic processes.
|
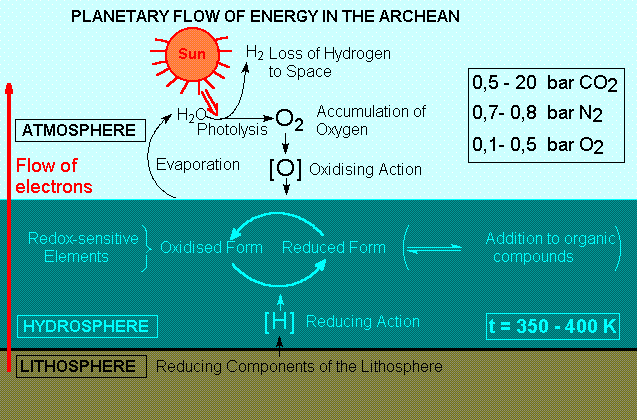
GEOCHEMICAL CYCLES OF THE
BIOGENIC ELEMENTS IN THE ARCHEAN.
|
The
span of electrochemical potentials in the protobiological
Earth is calculated to range from the values of
lithospheric divalent iron to those of atmospheric
oxygen.
The first geochemical cycles are made up from the
possible chemical species for the biogenic elements
and their transitions within this interval.
The oxidising nature of the primitive atmosphere
would be determined by the abiotic production of
oxygen, originating from the photolysis of atmospheric
water vapor followed by gravitational loss of hydrogen.
The sparingly low solubility of oxygen in the hot
primordial oceans, and the scarcity of emerged lands
would require the maintainance of a high concentration
of oxygen in the atmosphere, so that the flux of
electrons from the hydrospheric and lithospheric
compartments could compensate for the loss through
photolysis.
Most reducing gases originated from volcanic activity
would be delivered directly to the hydrosphere or
would have short residence in the atmosphere. Small
amounts of the less soluble methane and carbon monoxide,
however, could co-exist in equilibrium with the
oxidising atmosphere.
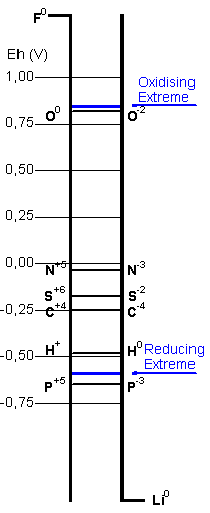
The stability areas for the chemical species derived
from biogenic elements are plotted in the Pourbaix
diagrams. The blue line links the extreme values
of pH and electrochemical potential calculated for
the hypercarbonic medium. It is intended to depict
, in an approximate view, the possible chemical
transitions in the proposed protobiological conditions.
In the case of sulfur the cycles would be further
complicated by the interactions with divalent iron.
The Pourbaix diagram for carbon includes just the
two extremes, the most reduced species - methane,
and the most oxidised, carbon dioxide (and its aqueous
derivatives, carbonic acid, bicarbonate and carbonate
ions), out of a great number of possible compounds
displaying intermediate reduction states.
|
|
| |

|
|
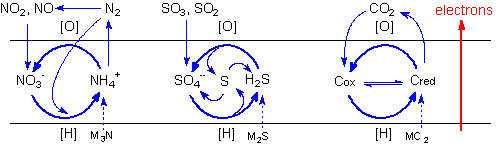
The carboxylating
and condensing properties imparted to the aqueous
medium (dubbed "hypercarbonic") by the
presence of a heavy CO2 atmosphere
are particularly important, since they permit carbon
fixation in the presence of reducing power, and
chiral propagation.
|
A HYPERCARBONIC WORLD.
| |
The
expression "hypercarbonic medium" describes
the reactive properties of the oceanic environment
of the early Earth under the atmospheric composition
postulated in the ecopoesis model. Owing to the
high amount of dissolved CO2,
chemical species such as carbonic acid, bicarbonate
and divalent ions (mainly the alkaline-earth magnesium)
are present in much higher concentrations than
in today's ocean, imparting carboxylating properties
which may affect amino groups and carbanions.
Under the thus arising carboxylation-decarboxylation
equilibrium, many organic compounds would form
series of closely related "hypercarbonic
analogs", whose members differ solely by
the amount of CO2 added
to their molecules.
The
highly polar medium with a lowered water activity
(Aw) governs the hydration-dehydration
equilibrium. Additionally, a reduction-oxydation
equilibrium is a part of the oceanic environment
as a whole, poised between a lithosphere delivering
reducing ions (mainly divalent iron and sulfur)
and the oxic atmosphere. The factors determining
the carboxylation-decarboxylation equilibrium and
the hydration-dehydration equilibrium are termed
permanent factors and are linked to very slightly
fluctuating conditions whereas the reduction-oxidation
equilibrium is governed by the so-called eventual
(contingent) factors, which vary widely, within
the redox extremes of the protobiological environment.
The chemistry of the hypercarbonic medium lies at
the heart of the ecopoesis model because it reconciles,
as dictated by the principle of congruence, the
biological relevance of carboxylation and decarboxylation
reactions with the geological and astronomical evidence
of a heavy CO2 paleoatmosphere.
|
|
Since carbon dioxide
is extremely stable towards reduction in aqueous
solutions, carbon fixation would depend on the carboxylation
of pre-existing carbon compounds. Protobiological
carbon cycles would run in the anabolic sense intercalating
carboxylation reactions of organic compounds with
reduction steps. Conversely, catabolic cycles would
alternate decarboxylations and oxidation steps,
as in modern metabolic cycles. The possibilities
of non-enzymatic analogues of such reactions in
the hypercarbonic medium are discussed to some detail
in the book, as well as the mechanisms through which
the reduction-oxidation equilibrium in the protobiological
ecosphere would be tightly bound to the carboxylation-decarboxylation
processes.
|
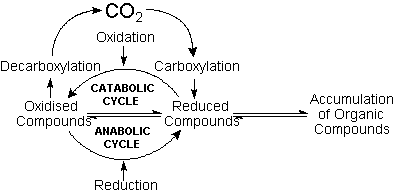
Carbon fixation
would also require at least a modest input (primary
input) of lithospheric carbide-derived hydrocarbons.
Acetylene, in particular, originated by the action
of water on divalent ionic carbides, could be directly
converted, through hydration and carboxylation,
into pyruvic and oxaloacetic acids, which are a
part of the very core of biochemical pathways. The
likelihood of this reaction suggests the bi-directional
Krebs cycle as the main feature of the circulation
of carbon in the early ecosphere.
The availability of reducing power for anabolic
processes (deriving mainly from lithospheric divalent
iron and sulfides), would, in most oceanic domains,
outbalance the total amount of oxidising power brought
in by the slightly soluble atmospheric oxygen, causing
the accumulation of organic compounds in the hydrosphere.
Inquiry into the nature of
these compounds and their formation processes may
be helped by the presumption of a congruent evolution.
|
THE PRINCIPLE OF
CONGRUENCE.
| |
The principle
of congruence is a heuristic tool meant to guide
the investigation of the history of living systems.
It states simply that modern life is a highly useful
and necessary reference in the study of life's previous
stages. Congruence can be envisioned as complement
to evolutionary theory since it represents the search
for evidences of common descent at a biochemical
level.
Although the biochemical similarities among existing
organisms are generally held as an unambiguous proof
of their common evolution, classical models for
the origin of life propose a previous, "prebiotic",
world, where organic compounds would have accumulated
through processes foreign to today's biochemistry.
The first theoretical propositions regarding the
research of the evolutionary nature of metabolism
were laid out by Granick in the 40s, and were later
applied, mainly by De Duve and by Morowitz, to the
field of the origin of life. The Ecopoesis model
further extends the realm of congruence, from the
evolution of organisms to that of the ecosphere
as a whole.
This conception implies that geochemical cycles
of biogenic elements have always been essentially
the same, but also that the steep energy gradients
which fuel vital processes have not been created
by organisms. Instead, the very existence of energy-yielding
processes in the primordial environment, especially
the aerobic degradation of organic matter (total
oxidation to CO2),
would have been essential to the creation of order
inherent to organismal life.
The use of the term "protobiological",
implying primitive life, as opposed to "prebiotic"
(before life) emphasizes the kind of continuity
which characterizes the principle of congruence
and is consistently followed in the ecopoesis model. |
|
Congruence requires
chemical analogy between these two stages of the
biochemical evolution , and the action of protobiological
environmental factors is shown to parallel that
of coenzymes.
|
CONGRUENT PROCESSES
OF THE HYPERCARBONIC WORLD .
| |
Reactive
factors of the environment and the corresponding
metabolic reactives.
1 - Permanent.
Environmental
factors (and opposite reaction) |
Corresponding
metabolic reactives |
Origin |
+ H2O
- H2O
|
(spontaneous)
ATP, GTP, etc. |
Hydroatmosphere
|
+ CO2
- CO2
|
Biotin
(spontaneous) |
Hydroatmosphere
|
2 -Eventual
(contingent) primary.
[H]
[O]
|
TH
- Reduced Transporters
(NADH, FADH2, etc.)
T - Oxidised
Transporters
(NAD+, FAD, etc.) |
Lithospheric
Reducing Power
Atmospheric Oxygen |
+
H2S
- H2S
(+H2O)
|
CoenzymeA-SH
(spontaneous) |
Hydroatmosphere
(S Cycle) |
+
NH3 (NH2COOH)
- NH3
(+H2O)
|
Amine
compounds
(spontaneous) |
Hydroatmosphere
(N Cycle) |
+
H2PO4-
- H2PO4-
(+H2O) |
ATP,
GTP, etc.
(spontaneous) |
Hydroatmosphere |
3 - Eventual
(contingent) secondary.
H2S2 |
Lipoic
acid |
H2S
/ [O] |
NH2COOH
/ [H]
(NH2COSH / [H])
Nitrogen nucleophile |
Pyridoxamine |
NH2COOH
/ [H] |
CN-
Carbon nucleophile |
Thiamine |
Cyanide
or Formic acid
derivatives (One-carbon) |
|
|
Coenzymes represent the first step towards the
incorporation of the primordial naked metabolism
into units bearing catalytic activity. That this
transition should have happened mainly during
the "RNA world", where metabolic functions
where carried out by ribozymes, is documented
by the fact that most of these coenzymes are structurally
related to ribose nucleosides.
Chiral propagation (enantioselective polymerization)
would ensure the synthesis of homochiral polypeptides
owing to the formation of prochiral centres in
the hypercarbonic derivatives of peptidic materials.
|
CHIRAL PROPAGATION IN THE
HYPERCARBONIC MEDIUM.
| |
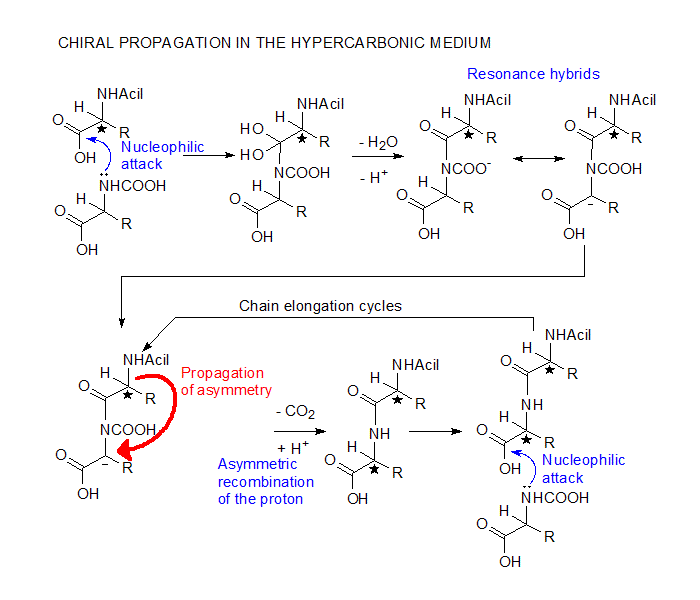
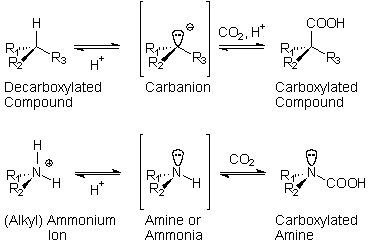
The
chiral propagation mechanism here shown for racemic
aminoacids is a minimally stripped down version
of the possible equilibrium forms in the hypercarbonic
medium. Accordingly, N-carboxylation and protonation
of the main-chain carboxyl are essential, but the
actual reactive species may involve the addition
of CO2 or H2S
to the peptidic material, with the use the corresponding
aminodiacids and thioacid derivatives. Although
modern organisms use optically active aminoacids,
this mechanism is otherwise fully congruent with
present-day biochemistry.
In the hypercarbonic medium, aminoacids are deprived
of their zwitterionic properties. The nucleophilic
attack of the carboxylated amino group on the protonated
carboxyl proceeds easily, generating an adduct which
contains a resonance stabilised carbanion. The asymmetric
recombination with the proton is oriented by the
aminoacid residue added in the previous cycle. Since
initiation may occur through acylation by an achiral
carboxylic acid, the first aminoacid of the chain
can be any of the two enantiomers. It will determine,
however, the chiral orientation of all its subsequents,
giving rise to homochiral polypeptides.
Another important aspect is that the acylation of
the amino group of the aminoacid residue raises
the pka of the carboxyl group
thereby increasing the proportion of protonated
carboxyls and favouring chain elongation rather
than the attack of monomers.
The mechanistic theory for the origin of chirality
in the biological world is an integral part of the
ecopoesis model. |
|
The organismal
characteristics of life are only gradually acquired
and cells are fairly late-comers in this model.
|
EVOLUTION OF ORGANISMALITY:
THE PROTOBIOLOGICAL HABITS.
| |
PROTOBIOLOGICAL MATERIALS
AND THE FLOCCULAR HABIT.

The initial aggregation
of organic matter (dubbed "floccular habit"),
countering the entropic tendency to dilution, is
ascribed to a combination of factors:
1) The lowered water
activity (Aw) of the hypercarbonic
medium.
2) The coalescence
of hydrophobic compounds (minimizing energy due
to charge-separation in the highly polar medium).
3) The kinetics
of formation of thio-acids and their partial extraction
into the lipid phase.
4) The stabilization
of the hydrophobic micelle through the formation
of surface amphiphilic compounds.
5) The facile formation
of peptide bonds due to the destruction of the zwitterionic
character of amino-acids through N-carboxylation.
6) The adhesion
of the peptide fraction to the micellar surface
amphiphilic compounds due to charge affinity, possibly
involving polyvalent cations.
7) The adsorption
of low-molecular weight organics to the peptide
fraction.
All these properties
would stabilize colloidal aggregates in dilute solution.
They are even compatible with the vision of the
early Earth as a "water-world", devoid
of significant evaporating surfaces.
The preferential solubility of oxygen in the lipid
phase favours oxidation processes next to the micelle
surface. A rather simple process, homogeneous phosphorylation
- analogous to substrate level phosphorylation,
requiring the oxidation of hydrogen sulphide to
hydrogen disulphide (lipoic acid analogs), would
cause the oxidation of a-ketoacids to thioacids,
permitting the synthesis of 'high-energy' compounds.
Sulfur-containing amino-acids in the peptide fraction
could bind divalent iron of lithospheric origin.
These primitive iron-sulfur clusters, ancestors
of today's ferredoxins, are responsible for the
localization of reducing power, concentrating redox
potentials within early colloidal aggregates, which
would thus have both reducing and oxidising poles.
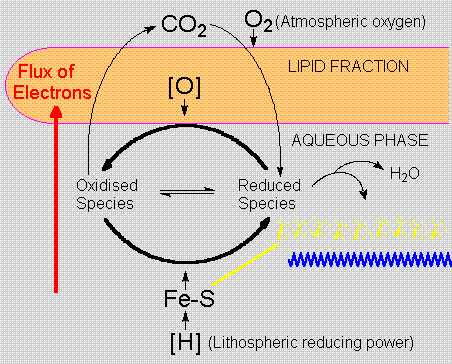
An initially loose and generic interaction between
polypeptides and phosphodiester polymers (PDEP-
or primitive RNA) would increase in specificity
giving rise to translation, and to structural and
functional control which characterises the "reticular
habit".
THE RETICULAR
HABIT.

Although heterogeneous
phosphorylation ("respiration") requires
physical barriers, like membranes, it could be developed
before the advent of the "cellular habit",
since protons resulting from the oxidation of lipid
soluble electron transporters would segregate in
protonic (or acidic) vacuoles, thus creating potential
between this vacuole and the external (or metabolic)
compartment, which would provide a more efficient
path for the synthesis of high-energy compounds.
CELLULARITY AND
CELLULAR RESPIRATION.
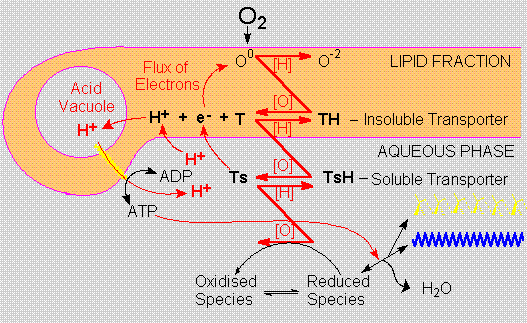
In the Ecopoesis
model cellularity does not arise as a method to
protect organic compounds from dilution, but to
promote and to profit from their oxidation. This
proposal of an aerobic dawn for cellularity is based
on the functional energetic properties of cells
rather than on their physical appearance.
CELLULAR HABITS.

Gradual
enclosure of the metabolic recess would lead to
the cellular habit (where the protonic vacuole is
no longer needed). |
|
THE BOOK.
 |
Click here
to download ECOPOESE - A criação da
ecosfera, 2ª Ed., 2006; (in portuguese) - Free
PDF (1.7 Mb, 345 pages).
Or leave a message to buy
the printed version.
This document is protected by copyright.
However, any parts of the work can be freely reproduced
or translated for non-commercial purposes, provided
the source is clearly referenced.
While quoting reference should
be made in the following form:
Félix de Sousa, Raul A., (2006). Ecopoese
- A criação da ecosfera, 2ª Ed.,
Rio de Janeiro. |
AN ARTICLE.
An abridged viXra version
in english is available for download here
( Free PDF 526 kb, 39 pages )
To cite:
Félix de Sousa, Raul A. (2010). The
Ecopoesis Model: Did Free Oxygen Fuel the Origin of Life?
viXra: 1010.0001 |